Neonatal Procedure Pack
BRIEF DESCRIPTION
Custom procedure pack designed for neonatal procedures
PRODUCT CODE
05-115343AA-03
COUNTRY OF ORIGIN
The Netherlands
TRADE NAME
KINDER SET
APPROVAL BY THE NATIONAL ORGANIZATION FOR MEDICINES (NOM)
2820007378388
KINDER SET is a complete, high quality neonatal procedure pack designed to meet the needs of the surgical site during neonatal procedures.
It is a ready to use solution that includes all essential components, contributing to reduced preparation time while maintaining full sterility throughout the process.
It is manufactured using high quality materials, sourced only from certified suppliers in the USA, the United Kingdom, and Europe. Final assembly and packaging take place in the Netherlands by Medica Europe B.V., in compliance with strict quality standards and the European Regulation EU 2017/745 (MDR).
Package Contents
KINDER SET is a complete neonatal procedure set that includes the following components:
|
S/N |
DESCRIPTION |
UNIT |
|---|---|---|
|
1 |
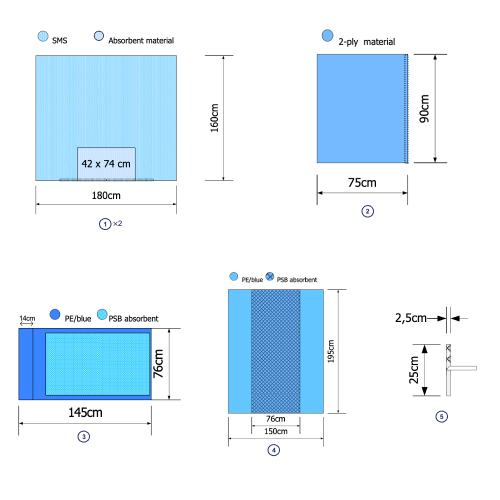
Cover SMS 180X160cm with impermeable, highly absorbent area 42X74cm, with hypoallergenic adhesive tape for children |
2 |
|
2 |
Side cover 2PLY 90X75cm, with hypoallergenic adhesive tape for children |
1 |
|
3 |
MAYO syrgical table cover 76X145cm |
1 |
|
4 |
Cover 195Χ150cm with impermeable, highly absorbent area 195Χ76cm |
1 |
|
5 |
Self-adhesive cable sheath tape - hypoallergenic for children 25X2,5cm |
1 |
Key Benefits
- CE MDR Compliance: Class IIa medical device in accordance with EU Regulation 2017/745
- Complete set: All necessary components in a single, integrated package
- Reduced preparation time: Ready to use solution for faster and more efficient setup
- Consistent high quality: Carefully selected consumables from internationally certified suppliers
- Optimized logistics: Simplifies inventory and procurement management
- Full traceability: LOT and Reference Number indicated on each package
Certifications & Packaging
The product is supplied:
- Sterile
- Single-use
and complies with the following standards:
- ISO 13485:2016
- EN ISO 13485:2016
- CE marking in accordance with EU Regulation 2017/745 (MDR)
 English
English Greek
Greek